David Pigott MD RDMS FACEP
Co-Director UAB Emergency Ultrasound
Case Presentation:
51M who denied PMH other than smoking presented with shortness of breath. He had been hospitalized 2 weeks prior at an OSH for “pneumonia and fluid in his lungs” but left AMA the next day. He says he was sent home on oral levofloxacin. He now reports 3 days of worsening dyspnea. On exam, the patient was afebrile, tachypneic, tachycardic to 110, with an oxygen sat of 95% on room air. Lungs were clear and cardiac exam was rapid and regular. No peripheral edema was noted and the patient was alert and nonfocal.
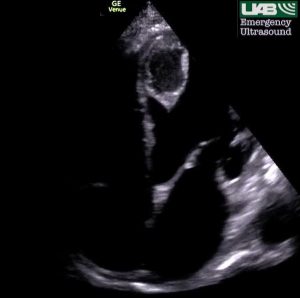
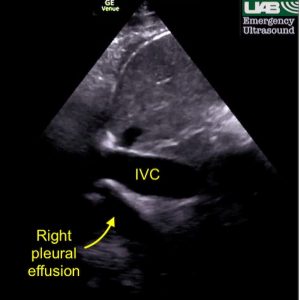
Copyright permission has been obtained from the UAB Department of Emergency Medicine Division of Ultrasound, Dr. David C. Pigott MD Co-Director of UAB EM Emergency Ultrasound
Limited bedside echo was markedly abnormal, demonstrating a poor LV ejection fraction as well as a large, cystic, mobile structure within the left ventricular chamber. A dilated IVC with minimal respiratory variation was noted as well as a right pleural effusion. The right pleural effusion appears as an anechoic fluid collection deep to the IVC on the typical longitudinal IVC view.
Clinical course:
The patient was placed on BiPAP on arrival due to significant dyspnea. Chest XR showed R pleural effusion with R basilar airspace opacities. EKG showed V3-V4 anterior ST elevation without reciprocal changes. Labs: hsTrop: 104, BNP >3000. Influenza and COVID tests were negative.
The patient was started on vancomycin and Zosyn for possible pneumonia, given IV Lasix and, after Cardiology fellow evaluation, admitted to the CCU on heparin drip.
Discussion:
The diagnosis of left ventricular thrombus is an important one, and is more likely to be present in patients with poor left ventricular function, and in the patients with anterior or apical MI, as the hypokinetic or akinetic left ventricle establishes an ideal location for thrombus to occur. LV thrombus typically occurs in the first 2 weeks following MI, affecting 4% to 10% of patients in the era of primary percutaneous coronary intervention.
If you recall Virchow’s triad, named after the famous Dr. Triad, stasis, endothelial injury and hypercoagulable state predispose patients to thrombus formation.
In this setting, LV thrombi can grow quite large in size and, as seen in this case, even undergo cystic degeneration, not unlike the gradual breakdown of a subcutaneous hematoma in the setting of trauma. While this pattern of a central hypoechoic or anechoic area within the thrombus is uncommon, it has been well described in the cardiology literature, although it has never been reported in the EM literature.
In the absence of systemic anticoagulation, the risk of arterial embolization in patients with left ventricular thrombus has been estimated at 10% to 15%. Most embolic events occur within 3 to 4 months after acute myocardial infarction. Anticoagulation has been shown to reduce the rate of embolization by 86%.
Follow-up:
The patient was admitted to the CCU, aggressively diuresed, and offered substance abuse counseling after it was revealed that he was an active methamphetamine user. Echocardiography confirmed the presence of multiple LV thrombi as well a left ventricular EF <20%. He was bridged from heparin to Loveonx/Warfarin and discharged 7 days later.
References:
- Ouf SG, Abualnaja S, Ghazal SN. A Large Left Ventricular Cystic Thrombus: Unusual Presentation of a Common Entity. CASE (Phila). 2017 Dec 19;1(6):216-220.
- Massey CV, Muller AL, Bennett BD, Gregory E, Alpert MA. Left ventricular pseudocyst caused by intraventricular thrombus: comparative echocardiographic and pathologic features. Am Heart J. 1994 Oct;128(4):831-3.
A good discussion of the development of LV thrombus after acute MI is available here:
Delewi R, Zijlstra F, Piek JJ. Left ventricular thrombus formation after acute myocardial infarction. Heart. 2012 Dec;98(23):1743-9.